Small Molecule Antagonist of the RAGE-DIAPH1 Signaling Axis for Diabetic Kidney Disease (DKD) Treatment
A new small molecule antagonist of the RAGE-DIAPH1 signaling axis, an important therapeutic node for the treatment of diabetes complications
Technology Overview
The inventors have developed a new, potent small molecule antagonist of the RAGE-DIAPH1 signaling axis, which is an important but underexplored therapeutic node for the treatment of diabetes complications, such as DKD. Presently, the inventors are working to further validate the compound’s efficacy in inflammation and wound healing assays, as well as to determine the compound’s pharmokinetics and target specificity.
In prior work (Manigrasso et al. Sci Transl Med 2021), an earlier generation RAGE-DIAPH1 antagonist (termed RAGE229) was developed using a “hit” scaffold identified from high-throughput screening (HTS). RAGE229 is a high-affinity competitive inhibitor and shown to mitigate short- and long-term complications of diabetes in a diabetic mouse models without lowering blood glucose levels. Moreover, RAGE229 administration to diabetic mice was found to reduce the concentration of pathological biomarkers (TNF-alpha, IL6 and CCL2/JE-MCP1) and other functional indices characteristic of DKD. In all, prior work with RAGE229 establishes that inhibition of the RAGE-DIAPH1 signaling axis can effectively mitigate diabetic complications, such as DKD, through the attenuation of inflammatory signaling and not through the modulation of blood sugar levels.
Current Drug Development Stage
Hit-to-Lead stage: Currently validating lead antagonist in efficacy studies (inflammation and wound healing assays); pharmokinetics and target specificity are also being determined.
Background
Diabetic kidney disease (DKD), also called diabetic nephropathy, is a type of chronic kidney disease caused by diabetes that results in variable degrees of kidney damage. Diabetes presently afflicts 34.2 million people and approximately 20-40% of all diabetic persons will develop DKD. The prevalence of diabetes and DKD is expected to increase sharply in the US and abroad over the next decade. While there are numerous glucose-lowering therapeutics in the clinic, including newer classes of anti-diabetes agents, critical gaps exist in our therapeutic arsenal for generalizable disease-modifiying treatments of diabetic complications, such as DKD, arising from either Type 1 or Type 2 diabetes.
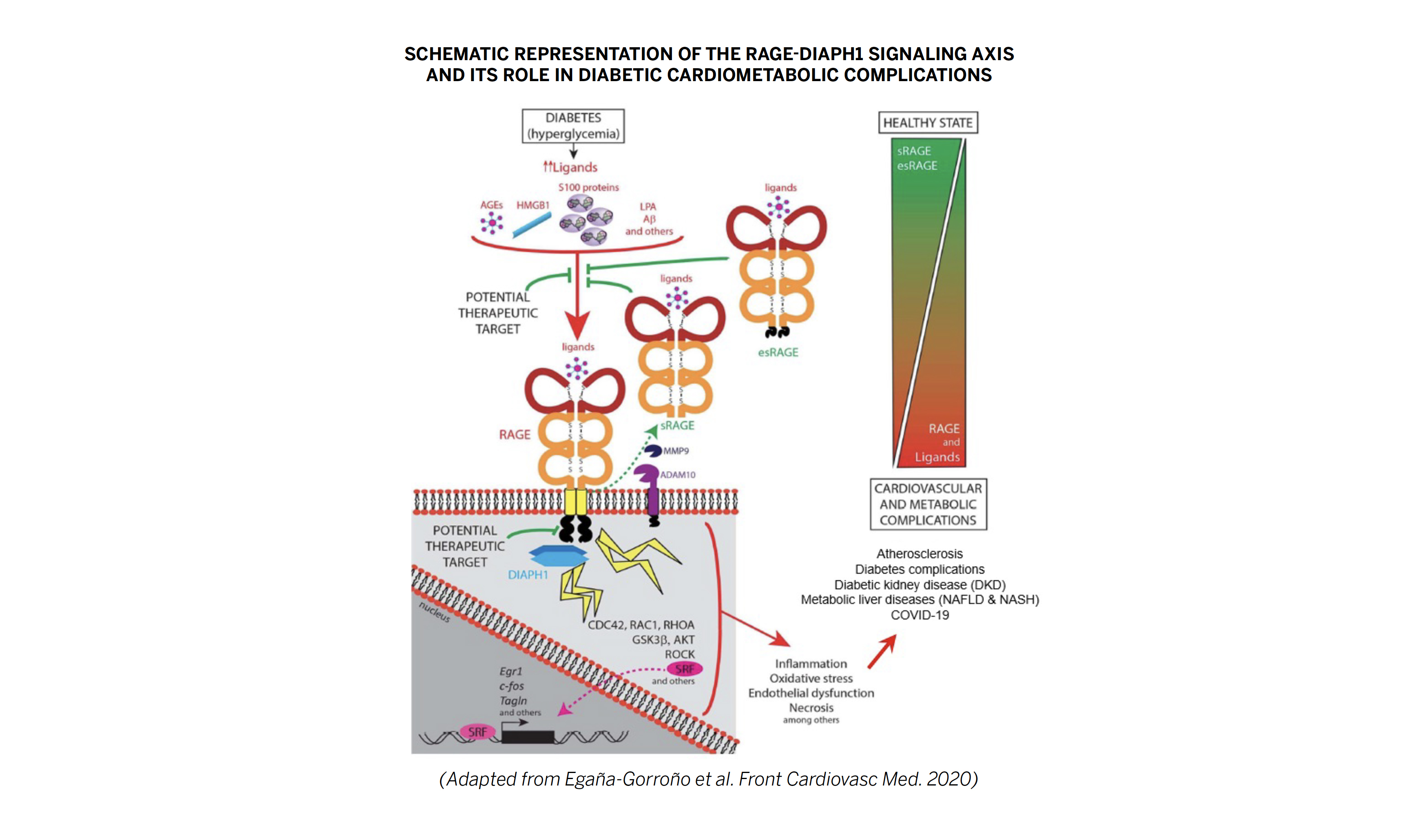
The RAGE signaling pathway (see pg. 2 schematic) is a promising therapeutic target for the treatment of diabetes complications, such as DKD. The Receptor for Advanced Glycation End Products (RAGE) binds and transduces signals from diverse array of ligands, including advanced glycation end products (AGEs) and proinflammatory ligands that accumulate in diabetic tissues. Activation of RAGE triggers intracellular signaling cascades mediated by the downstream effector DIAPH1 leading to pathological cellular responses (inflammation, necrosis, oxidative stress, etc.) and subsequent cardiometabolic complications. Therefore, pharmacological inhibition of the RAGE:DIAPH1 signaling axis is a promising therapeutic strategy for the treatment of diabetes complications, such as DKD.
Applications
- Treatment of diabetes complications, including:
- Impaired wound healing
- Diabetic kidney disease (DKD)
- Potential treatment of other indications where inhibition of RAGE signaling shows some preliminary benefits
- Atherosclerosis
- Non-alcoholic fatty liver disease (NAFLD)
- Non-alcoholic steatohepatitis (NASH)
- COVID-19
Advantages
- Unique therapeutic node
- Inhibition of the RAGE-DIAPH1 signaling axis attenuates pathological inflammatory signaling without lowering blood glucose levels
- Well-characterized, disease-implicated signaling pathway
- The RAGE signaling pathway is a well-known driver of proinflammatory responses in cardiometabolic complications
- Mode of antagonist binding
- Prior structural studies with RAGE229 suggest new antagonist to bind RAGE at the RAGE:DIAPH1 interaction interface
- Generalizable therapeutic across diabetes subtypes
- RAGE signaling antagonists can treat diabetes complications, such as DKD, arising from both Type 1 and Type 2 diabetes subtypes
- Method of compound administration
- Prior data for RAGE229 suggest efficacy using both topical and oral/medicated chow approaches
Opportunity
Seeking a partnership with a small-to-large biopharma company in the inflammatory/diabetes-associated disease space to in-license the technology.
Intellectual Property
NYU has filed a PCT application covering the composition of the lead RAGE:DIAPH1 inhibitor and its method of use.
Additionally, NYU holds patents and patent applications covering the parent RAGE:DIAPH1 inhibitor series (to which the current lead compound was derivatized) and other backup series.
-
swap_vertical_circlemode_editAuthors (1)Ann Marie Schmidt, MD
-
swap_vertical_circlelibrary_booksReferences (1)
- Ann Marie Schmidt, MD, et al. , Small-molecule antagonism of the interaction of the RAGE cytoplasmic domain with DIAPH1 reduces diabetic complications in mice
-
swap_vertical_circlecloud_downloadSupporting documents (2)Product brochureSmall Molecule Antagonist of the RAGE-DIAPH1 Signaling Axis for Diabetic Kidney Disease (DKD) Treatment.pdfMarketing BriefNYU_RAGE inhibitors_MarketingBrief - SCH08-06.pdf (190 KB)DOWNLOAD